
(b) In order to compute the atomic radius we must first determine the lattice parameter, a, using Equation 3.14, and then R from Equation 3.1 since Ir has an FCC crystal structure. 
Solution (a) From the data given in the problem, and realizing that 69.20° = 2θ, the interplanar spacing for the (220) set of planes for iridium may be computed using Equation 3.13 as If the angle of diffraction for the (220) set of planes occurs at 69.20° (first-order reflection) when monochromatic x-radiation having a wavelength of 0.154 nm is used, compute (a) the interplanar spacing for this set of planes, and (b) the atomic radius for an iridium atom. 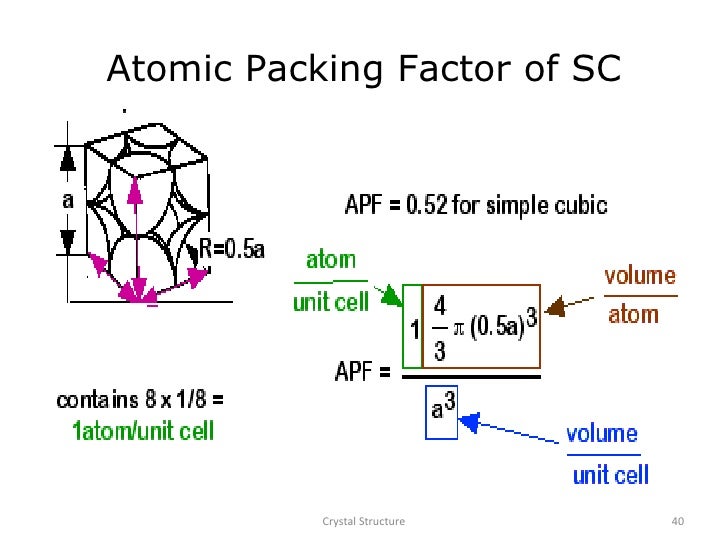
But a depends on R according to Equation 3.3, and ⎛ 4R ⎞3 64 R 3 VC = ⎜ ⎟ = ⎝ 3⎠ 3 3ģ.61 The metal iridium has an FCC crystal structure. Since there are two spheres associated with each unit cell for BCC ⎛ 4π R 3 ⎞ 8π R 3 VS = 2 (sphere volume) = 2 ⎜ ⎟= 3 ⎝ 3 ⎠Īlso, the unit cell has cubic symmetry, that is VC = a3. Solution The atomic packing factor is defined as the ratio of sphere volume to the total unit cell Show that the atomic packing factor for BCC is 0.68.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
